C. Scott Hartley* and Jian He
J. Org. Chem. 2010, 75, 8627–8636
[Published version]
Abstract
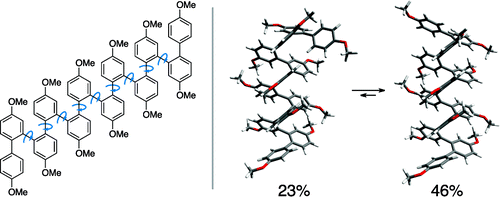
The o-phenylenes represent a fundamental class of conjugated polymers that, unlike the isomeric p-phenylenes, should exhibit rich conformational behavior. Recently, we reported the synthesis and characterization of a series of o-phenylene oligomers featuring unusual electronic properties, including surprisingly long-range delocalization as measured by UV−vis spectroscopy and hypsochromic shifts in fluorescence maxima with increasing length. To rationalize these properties, we hypothesized that the oligomers predominantly assume a stacked helical conformation in solution. This assertion, however, was supported by only indirect evidence. Here we present a thorough investigation of the conformational behavior of this series of o-phenylenes by dynamic NMR spectroscopy and computational chemistry. EXSY experiments, in combination with other two-dimensional NMR techniques, provided full 1H chemical shift assignments for at least the two most prevalent conformers for each member of the series (hexamer to dodecamer). GIAO density functional theory calculations were then used to relate the NMR data to specific molecular geometries. We have found that the o-phenylenes do indeed assume stacked helical conformations with disorder occurring at the ends. Thus, the o-phenylene motif appears to have great potential as a means to organize arenes into predictable three-dimensional arrangements. Our results also illustrate the power of 1H NMR GIAO predictions in the solution-phase conformational analysis of oligomers, particularly those with a high density of aromatic subunits.
